
First Detection of Red Leaf Blotch: A New Disease of Almond in California
Red leaf blotch (RLB), caused by the fungal pathogen Polystigma amygdalinum, is one of the most important leaf…
Read ArticleArticle Archive

Red leaf blotch (RLB), caused by the fungal pathogen Polystigma amygdalinum, is one of the most important leaf…
Read Article
As the agriculture industry continues to evolve, staying informed and connected is more critical than ever. The Crop…
Read Article
Barn owls (Tyto alba) are birds of prey known for their heart-shaped faces and eerie, yet captivating, screeches…
Read Article
Intensified drought in California has limited groundwater supply for crop production. Therefore, vegetable growers need to implement more…
Read Article
Roundup Ready technology builds genetic resistance to glyphosate into crops, providing an excellent tool for weed management. Initial…
Read Article
Crop yields are often lower in organic production systems compared to conventional farming systems (De Ponti et al.…
Read Article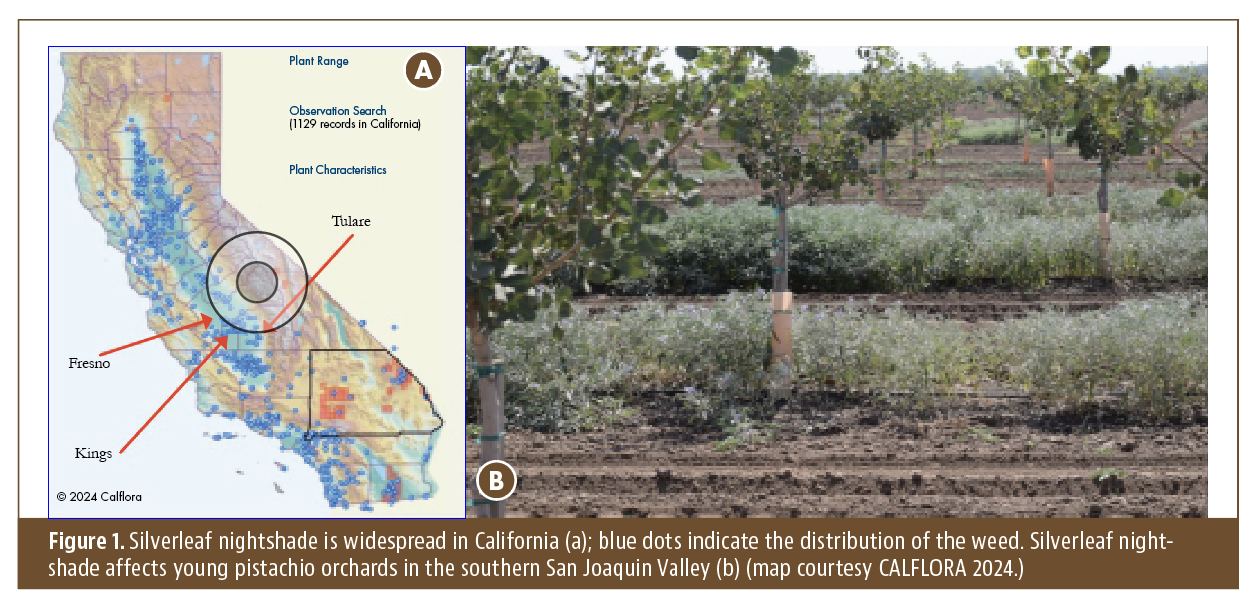
Silverleaf nightshade, Solanum elaeagnifolium, is a perennial weed native to South America, Mexico and the southwestern and southern…
Read Article
Growers in the west are facing more and more pressure to account for their water use. On May…
Read Article
Accurate nitrogen monitoring and appropriate nitrogen management are crucial in California due to the state’s unique environmental challenges…
Read Article
In the world of agricultural sales, particularly within the rich and varied landscapes of California, understanding and overcoming…
Read Article