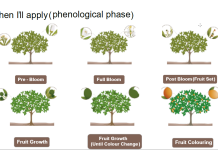
The goal of properly timed foliar-applied fertilizer is to increase the economic benefit derived from the grower’s fertilization program. In this strategy, fertilizer is applied at key stages in citrus tree phenology (the series of developmental events that result in fruit production and tree growth) (Figure 1). Key stages of tree phenology are associated with important physiological and developmental processes. The fertilizer application is timed to stimulate a specific physiological process and achieve a plant growth regulator effect that increases flowering, fruit set, yield, fruit size or fruit quality, even when the tree is not deficient in the applied nutrient based on standard leaf or other tissue analyses. Properly timing the fertilizer application is important because the developmental stage of the target organ determines the result obtained.

Winter Applications Increase Yield and Fruit Size
The key stage of citrus tree phenology being targeted is bud determinacy, the irreversible commitment of buds that transitioned from vegetative shoot development to floral development in the fall to produce inflorescences and flowers. When a bud is determined, it can no longer revert to vegetative shoot growth. At the microscopic level, bud determinacy is identified by the initiation of sepals, the green outer leaves that cover and protect the developing floral bud (Lord and Eckard 1987). Due to annual variations in temperatures and the fact that not all shoots and buds are the same age or at the same stage of development, January 1 through February 15 is an effective window within which to make foliar applications of urea or potassium phosphite to increase flowering in citrus.
Low-biuret urea (46% N, 0.25% biuret, 25 lb N/acre or a 50 lb bag of low-biuret urea per acre) applied in mid-January to target irreversible commitment to flowering in ‘Washington’ navel orange trees resulted in a net increase in three-year cumulative yield of 17,355 lb/96 trees/acre compared to control trees receiving five-fold more urea applied to the soil. Delaying the foliar-application of low-biuret urea to mid-February resulted in a net increase in three-year cumulative yield of 13,757 lb/96 trees/acre. In both cases, yield of commercially valuable size fruit of packing carton sizes 88+72+56 (2.7 to 3.5 inches in transverse diameter) increased as total yield increased (Ali and Lovatt 1994). Similarly, potassium phosphite as Nutri-Phite (0-28-26; Verdesian Life Sciences), which supplies P as PO3, not PO4, applied at 0.64 gal/acre in January to target irreversible commitment to flowering of Valencia orange trees in Florida resulted in an average net increase of 900 inflorescences per 12-inch square frame, a 166% increase in inflorescence number compared to untreated control trees, and produced a net increase in yield of 6,853 lb/acre (Albrigo 1991). In a second experiment, foliar-applied Nutri-Phite (0-28-26, 0.64 gal/acre) in January resulted in a net increase in four-year cumulative Valencia orange yield of 25,247 lb/acre, an average net increase of 6,311 lb/acre/year for the four years of the experiment, with an average net increase of more than 400 lb total soluble solids per acre per year (Albrigo 1991).
Summer Applications Increase Yield
The key stage of phenology being targeted is maximum peel thickness, which marks the end of Stage I of fruit development in citrus in which fruit growth is predominantly by cell division, and the beginning of Stage II of citrus fruit development, the period of exponential fruit growth by cell expansion. At maximum peel thickness, all the cells that make up the mature fruit are present. Subsequent fruit growth is by the uptake of water into the juice sacs. The cells of the albedo, the white layer of the peel, simply stretch to accommodate the increasing size of the juice sacs; only the flavedo, the outer colored layer of the peel, continues cell division through harvest. The goal is to stimulate additional cell divisions just prior to maximum peel thickness. One additional cell division would double the number of cells in the fruit, a second cell division would quadruple the number of cells, etc. When these additional cells enlarge, fruit size is increased. Field research with navel orange, Valencia and mandarin cultivars documented the efficacy of foliar applications of low-biuret urea or potassium phosphite made between the last week of June and mid-July.
Foliar-applied low-biuret urea (46% N, 0.25% biuret, 25 lb N/acre) in early July to target maximum peel thickness in ‘Washington’ navel orange trees resulted in a net increase in yield of commercially valuable size fruit of packing carton sizes 88+72 (2.7-3.15 inches in transverse diameter) of 4,927 lb/109 trees/acre/year and a net increase in yield of larger fruit of packing carton size 56 (3.2 to 3.5 inches in transverse diameter) of 3,374 lb/109 trees/acre/year compared to untreated control trees (Lovatt 1999).
To stimulate cell division prior to maximum peel thickness, Nutri-Phite was applied at 0.49 gal/acre in mid-May and again in mid-July. Nutri-Phite produced a net increase in fruit of packing carton sizes 88+72 of 5,078 lb/109 trees/acre/year with a net increase in yield of larger fruit of packing carton size 56 of 4,158 lb/109 trees/acre/year. The Nutri-Phite treatment also increased fruit total soluble solids (TSS) by early November compared to the untreated control (P < 0.001), achieving a TSS:acid ratio of 8.1 by early November compared to 7.2 for fruit from untreated control trees (P < 0.01) (Lovatt 1999).
Application and Flowering Physiologies
It is well known that flowering in citrus is induced by low temperature (LT ≤59 degrees F day/ ≤50 degrees F, but >7 degrees F night) and by water-deficit stress (WD ≤-2.4 MPa stem water potential) for eight weeks. Inflorescence and flower number both increase as the duration of the LT or WD period increases. Citrus is unique in the plant world. Not only does flowering increase with the increased duration of WD, but also with the increasing severity of WD (Figure 2).
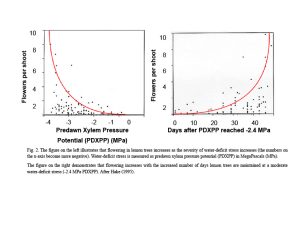
In the late 80s, my lab discovered that leaf ammonium concentrations increased with the increased duration of both LT and WD and that leaf ammonium concentrations increased in parallel with the increase in both inflorescence number and flower number in response to the duration of LT or WD (Lovatt et al. 1988a, b). Since urea applied to leaves is catabolized by the plant enzyme urease to ammonia and carbon dioxide (NH3 and CO2), my lab tested the capacity of foliar-applied urea to supplement ammonium accumulation during LT and WD and increase citrus flowering. For navel orange trees exposed to a 50% or 25% shorter period of low temperature, foliar-applied low-biuret urea increased flowering 194% and 230%, respectively. Foliar-application of low-biuret urea to lemon trees maintained at a moderate WD (-2.4 MPa predawn xylem pressure potential) at the end of the 50-day stress period increased flowering 260% compared to WD-treated trees not receiving foliar-applied low-biuret urea (Lovatt et al. 1988a, b). At the level of gene transcription, new evidence suggests that LT and WD initiate flowering through overlapping genetic pathways (Tang and Lovatt 2022). Thus, WD provides a tool to increase citrus flowering in growing areas experiencing warmer, dry winters, with foliar-applied low-biuret urea or potassium phosphite able to supplement both LT and WD to increase citrus flowering.
These Different Molecules Share the Same Benefits, but How?
Recent evidence suggests that both urea and phosphite increase tree nitrogen status and cytokinin biosynthesis. In both roots and leaves, nitrate and ammonium upregulate the expression of the key gene regulating cytokinin biosynthesis, IPT, the gene encoding isopentenyl transferase, which catalyzes the rate-limiting step in cytokinin biosynthesis (Sakakibara 2006; Sakakibara et al. 2006). Nitrate taken up by roots and cytokinin synthesized in the roots move in the xylem to the shoots and leaves, where nitrate upregulates the genes for N assimilation and the IPT gene for cytokinin biosynthesis. Cytokinin and metabolites synthesized in the leaves are then transported in the phloem to the roots. Thus, N and cytokinins work together to promote and coordinate root and shoot growth, bud break and flowering. Ammonium derived from foliar-applied urea would stimulate N assimilation and upregulate cytokinin biosynthesis. Cytokinins are known to promote flowering and fruit growth.
Surprisingly, phosphite was recently demonstrated to upregulate key genes for N assimilation, resulting in increased nitrate uptake (Vereet et al. 2021) and, as predicted by the information presented above, phosphite increased cytokinin biosynthesis (Swarup et al. 2020). Phosphite supplied to roots resulted in significantly greater cytokinin concentrations one day after phosphite treatment (P ≤0.05). Root cytokinin concentrations continued to increase during the week-long experiment to a level greater than untreated control plants (P ≤0.05).
The Window for Increasing Flowering is Now
Citrus flowering is induced by LT and WD stress. WD stress of approximately -2.4 MPa stem water potential can be maintained by deficit-irrigation, and thus WD can be used to supplement LT in citrus areas with warmer, dryer winters due to global climate change. Winter prebloom foliar-applied urea and Nutri-Phite can be used to supplement LT and WD stress to increase citrus flowering and yield. In addition, summer foliar-applied low-biuret urea or Nutri-Phite can be used at maximum peel thickness to further increase fruit size and yield of commercially valuable size fruit. In light of recent research results, foliar-applied urea and Nutri-Phite likely achieve a plant growth regulator effect by increasing N assimilation and cytokinin biosynthesis.
Foliar fertilizers should be applied in sufficient water for good canopy coverage (at a final pH of 5.5 +/- 0.5), but not to run off, which is a waste of product. Pooling of fertilizers at the leaf tip can result in tip burn. The application should be like a pesticide spray with good canopy mixing and coverage of the upper and under surfaces of the leaves on the exterior and interior of the canopy and the target organ. Always follow the product label. Results cited for properly timed foliar-applied potassium phosphite reported herein are for Nutri-Phite, Verdesian Life Sciences, the only commercial product for which results appear in peer-reviewed journals.
References
Albrigo, L.G. 1991. Effects of foliar applications of urea or Nutri-Phite on flowering and yields of Valencia orange trees. Florida State Hort. Soc. 112:1-4.
Ali, A. and Lovatt, C.J. 1994. Winter application of low-biuret urea to the foliage of ‘Washington’ navel orange increased yield J. Amer. Soc. Hort. Sci. 119:1144-1150.
Hake, K.D. 1995. Regulation of Flowering in Citrus limon by water-deficit stress and nitrogen compounds. PhD Dissertation, University of California, Riverside. 149p.
Lord, E.M. and Eckard, K.J. 1987. Shoot development in Citrus sinensis L. (Washington navel orange). II. Alteration of developmental fate of flowering shoots after GA3 treatment. Bot. Gaz. 148:17–22
Lovatt, C.J. 1999. Timing citrus and avocado foliar nutrient applications to increase fruit set and size. HortTechnology 9:607-612.
Lovatt, C.J., Zheng, Y. and Hake, K.D. 1988a. A new look at the Kraus Kraybill hypothesis and flowering in Citrus. 6th. Intl. Citrus Congr. 1:475-483.
Lovatt, C.J., Zheng, Y. and Hake, K.D. 1988b. Demonstration of a change in nitrogen metabolism essential to floral induction in Citrus. Israel J. Bot. 37:181-188.
Sakakibara, H. 2006. Cytokinins: Activity, biosynthesis, and translocation. Annu. Rev. Plant Biol. 57:431-449.
Sakakibara, H. Takei, K. and Hirose, N. 2006. Interactions between nitrogen and cytokinin in the regulation of metabolism and development. Trends in Plant Science 11: 40-448.
Swarup, R., Mohammed, U., Davis, J. and Rossall, S. 2020. Role of phosphite in plant growth and development. White paper, School of Biosciences, Univ. Nottingham.
Tang, L. and Lovatt, C.J. 2022. Effects of water-deficit stress and gibberellic acid on floral gene expression and floral determinacy in ‘Washington’ navel orange. J. Amer. Soc. Hort. Sci. 147(4):183-195.
Verreet, J.-A., Prahl, K.C., Loof, S. Birr, T. Klink, H., Cai, D. Xu, S. 2021. Phosphite plant biostimulant mode of action: gene expression, phytohormone levels, enzyme activity. Electronic Biological Inorganic Chemistry PO3 Workshop. (Christian-Albrechts-University of Kiel, Germany).