Listen to the audio version of this article (generated by A.I.)
For pest control advisors working in vegetable crops, few insects inspire as much frustration as diamondback moth (Plutella xylostella). Despite decades of insecticide development, this pest continues to evolve resistance, exploit production gaps, and resurge when control programs falter. As regulatory pressure increases and broad-spectrum options continue to erode, interest in the development of biological pest management (bioinsecticides, natural enemies, etc.) for this pest has grown. (Fig. 1)
Codling moth (Cydia pomonella) control in apples provides an amazing biological insect pest management success story. At first glance, application of this success story to diamondback moth may seem difficult. Codling moth is a tree fruit pest, diamondback moth attacks annual vegetable crops. And while their biology differs in important ways, codling moth remains one of the clearest examples of the investments needed to develop a durable, biologically based pest management system.

The central lesson from codling moth is not about a single tool or product. It is about process. Codling moth biological management did not emerge overnight. It was built slowly and deliberately. That history provides a realistic lens through which to evaluate what biological pest management for diamondback moth might look like, the scale of investment needed, and where advisors can play a critical role.
Two Very Different Ways to Manage Insects
For most of the twentieth century, pest management followed a broad-spectrum paradigm. Insecticides such as organophosphates, carbamates, and pyrethroids worked across many insect pests because they disrupted conserved physiological pathways, were residual contact-active, and had long residuals. Thus, one product could suppress multiple pests, often across several life stages. For managers, this meant fewer decisions, simpler timing, and predictable short-term results.
Diamondback moth fit well within this paradigm until it didn’t. Resistance to many if not most modes of action is now widespread globally (Zalucki et al. 2012; Furlong et al. 2013). The result has been increasing spray frequency, shrinking windows of efficacy, and rising production risk.

Biological pest management represents a fundamentally different approach. Rather than suppressing pests through generalized toxicity, biological systems exploit species-specific vulnerabilities: phenology, mating behavior, host specialization, microbial susceptibility, and ecological interactions. This approach is slower to develop, harder to generalize, and far more knowledge intensive. The payoff, when successful, is durability.
Codling moth illustrates this tradeoff better than almost any other agricultural insect pest.
Why Codling Moth Became a Biological Management Success
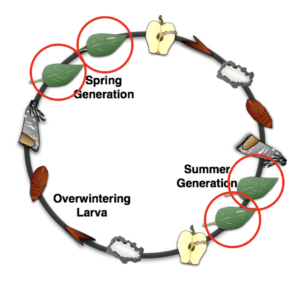
Codling moth (Cydia pomonella) is the most economically significant insect pest of apples. Its larvae feed internally within fruit, making damage severe and difficult to prevent. Historically, this biology forced growers to rely heavily on insecticides and precise spray timing.
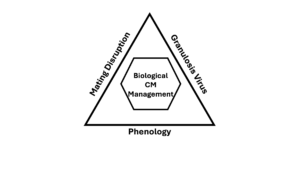
These challenges drove an extraordinary research and extension effort. Because codling moth control failed so easily when mistimed, researchers were compelled to understand the insect in detail. Over decades, this work coalesced into a biological management system built around three interacting components (Fig. 2).
The Codling Moth Biological Management Triangle
1. Phenology as the management framework
2. Mating disruption as baseline population suppression
3. Granulosis virus as targeted larval control
No single component works reliably on its own. Together, they form a system that has enabled large-scale organic apple production and reduced insecticide reliance across conventional orchards (Jones et al. 2008; Witzgall et al. 2010).
Phenology: The Foundation That Came First
The first pillar of codling moth biological management is phenology, and it predates biological products by decades.
A 40-Year Head Start
Codling moth phenology research began in the 1930s, initially as descriptive life-history work documenting instars, seasonal flight patterns, and overwintering behavior (Shelford 1927; Glenn 1922). By the 1950s and 1960s, researchers recognized that codling moth development was tightly temperature-driven and synchronized with host fruit phenology.
This led to the development of degree-day models, with the first widely adopted codling moth degree-day model published in the early 1970s (Riedl et al. 1976). By the 1980s, these models were being disseminated through extension programs across North America.
Estimated investment:
• ~40 years of research (1930s–1970s)
• 15–25 university and government programs
• 50–100 core scientists
• Hundreds of graduate students and technicians
• Dozens of extension professionals
For advisors, phenology transformed codling moth management from calendar spraying to event-based decision-making. Eggs and neonates became management targets, not just “the season.” (Fig. 3)
Mating Disruption: From Curiosity to Keystone Technology
Mating disruption, now considered the cornerstone of codling moth biological management, took decades to reach this status.

The concept was first proposed in 1967, with early demonstrations in cabbage looper (Shorey and Gaston 1967). Codling moth pheromone chemistry was elucidated in 1971 during a period of rapid growth in chemical ecology (Roelofs et al. 1971).
Commercial adoption, however, lagged far behind discovery. Economical pheromone synthesis, dispenser development, and field-scale validation took time. Widespread adoption did not begin until the 1990s, and area-wide programs only became common after 2000 (Cardé and Minks 1995; Witzgall et al. 2010).
By 2015, approximately 90% of Washington apple acreage was under mating disruption (Washington Tree Fruit Research Commission data).
Estimated investment:
• ~45–50 years (1970s–2010s)
• 20–30 research programs
• 75–150 scientists
• Hundreds of graduate students and postdocs
• Significant private-sector R&D
• Continuous extension involvement
For managers, mating disruption requires a mindset shift. It is prophylactic, not curative. It prevents future populations rather than killing existing ones. Its benefits accumulate over time, especially when deployed across large contiguous areas. If the tactic is removed from a program, codling moth rebounds and within a few seasons can become a serious problem once more.
Granulosis Virus: Precision with Constraints
The third pillar of codling moth biological management is codling moth granulosis virus (CpGV).
The virus was first isolated in 1963 (Huber 1963). European registration followed in 1984, U.S. registration in 1995, and widespread adoption occurred only after 2005. Development was constrained by challenges in mass production, UV stability, and application strategy (Lacey et al. 2008).
‘For diamondback moth, success is highly unlikely from a single new product.’
Advisors learned several hard lessons:
• CpGV must be ingested, so spray coverage is critical
• UV degradation limits residual activity
• Frequent low-rate applications outperform infrequent high-rate sprays
• Resistance can develop, but strain variation allows adaptation (Asser-Kaiser et al. 2007)
Estimated investment:
• ~40 years
• 15–20 research institutions
• 40–80 scientists
• Hundreds of graduate students and technicians
Why This Matters for Diamondback Moth
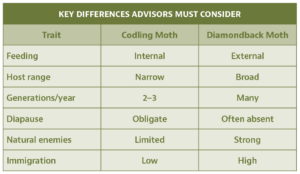
Diamondback moth (Plutella xylostella) presents a very different biological challenge, and different opportunities.

Diamondback moth’s rapid generation time and continuous host availability complicate suppression. However, unlike codling moth, diamondback moth larvae are exposed throughout development, providing better opportunities for control using insecticides and natural enemies (Fig. 4). For example, parasitic wasps such as Diadegma insulare can exert substantial control when broad-spectrum insecticides are minimized (Talekar and Shelton 1993; Furlong et al. 2013) (Fig. 5).
Adapting the Triangle for Diamondback Moth
The codling moth triangle cannot be copied directly, but it can be adapted. (Fig. 6).
A Diamondback Moth Biological Management Triangle
1. Phenology and population modeling
2. Mating disruption for baseline suppression
3. Biological insecticides and natural enemy conservation
Fortunately, we already have phenological models for diamondback moth. Seminal work by Harcourt established temperature-dependent development rates, which were later refined into degree-day models used for timing scouting and interventions in pest management programs (Harcourt 1957; Shelton et al. 1983; Kfir 1997). Application of these models may be especially useful during early season plantings while landscape populations of DBM are building.
Sprayable mating disruption products exist for diamondback moth but require frequent application. Point source dispensers have been developed, but optimal release rates, point-source density, and treated areas remain open questions. Annual cropping systems may offer opportunities for mechanized deployment that tree fruit systems never had. For example, dispensers could be integrated into plug trays or deployed using existing robotic weeding platforms.
Microbial tools such as Bacillus thuringiensis remain effective but are vulnerable to resistance without system-level integration. More recently, efforts have been made to commercialize DBM-specific granulosis virus, but optimal rates and timings of applications are still under development. Two such products are Lepigen (registered with both CA DPR and EPA) and Plutex (lacks EPA registration at time of writing).
The Big Lesson for Advisors
The most important lesson from codling moth is not about products. It is about expectations.
Codling moth biological management required:
• 70–90 years of cumulative research
• Dozens of institutions
• Thousands of people
• Continuous refinement
Biological pest management is built one insect at a time. Broad-spectrum chemistry hides biological complexity. Biological systems must confront it directly.
For diamondback moth, success is highly unlikely from a single new product. It will come from coordinated investment in phenology, behavior, microbial tools, and natural enemy conservation and from advisors willing to guide growers through a longer transition.


Looking Forward
Diamondback moth is unlikely to be “solved” quickly. The codling moth experience, however, shows that durable biological management is possible when the agricultural community commits to understanding an insect deeply enough to manage it predictably. Fortunately, we already have much of the baseline knowledge about the pest, and a variety of biological approaches exist, so a solution should not require 50 years of development. Instead, we can move directly to the integration phase of program development. Development of this program, however, will require a substantial investment in applied research and extension.
For PCAs and CCAs, this represents both a challenge and an opportunity. Those who understand the biology behind the tools will be best positioned to help growers navigate resistance, regulation, and the future of sustainable crop production.
Selected References
Asser-Kaiser, S., Fritsch, E., Undorf-Spahn, K., Kienzle, J., Eberle, K. E., Gund, N. A., Reineke, A., Zebitz, C. P. W., Heckel, D. G., and Jehle, J. A. 2007. Rapid emergence of baculovirus resistance in codling moth (Cydia pomonella). Journal of Invertebrate Pathology 95: 59–66.
Cardé, R. T., and Minks, A. K. 1995. Control of moth pests by mating disruption: successes and constraints. Annual Review of Entomology 40: 559–585.
Furlong, M. J., Wright, D. J., and Dosdall, L. M. 2013. Diamondback moth ecology and management: problems, progress, and prospects. Annual Review of Entomology 58: 517–541.
Harcourt, D. G. 1957. The development and use of life tables in the study of natural insect populations. Canadian Journal of Zoology 35: 343–378.
Jones, V. P., Brunner, J. F., Grove, G. G., Petit, B., Tangren, G. V., and Jones, W. E. 2008. Codling moth (Cydia pomonella) integrated pest management in apple orchards. Pest Management Science 64: 1157–1167.
Kfir, R. 1997. Biology and management of diamondback moth (Plutella xylostella). Annual Review of Entomology 42: 347–372.
Lacey, L. A., Thomson, D., Vincent, C., and Arthurs, S. P. 2008. Codling moth granulovirus: a comprehensive review. Biological Control 44: 221–238.
Roelofs, W. L., Comeau, A., Selle, R., and Riedl, H. 1971. Sex pheromone of the codling moth. Nature 233: 496–497.
Riedl, H., Croft, B. A., and Howitt, A. J. 1976. Forecasting codling moth phenology based on pheromone trap catches and physiological time models. Environmental Entomology 5: 121–127.
Shelton, A. M., Scriber, J. M., and Andow, D. A. 1983. Monitoring of diamondback moth populations using pheromone traps and degree-day accumulations. Journal of Economic Entomology 76: 135–141.
Talekar, N. S., and Shelton, A. M. 1993. Biology, ecology, and management of the diamondback moth. Annual Review of Entomology 38: 275–301.
Witzgall, P., Stelinski, L., Gut, L., and Thomson, D. 2010. Codling moth management and chemical ecology. Journal of Chemical Ecology 36: 80–91.
Zalucki, M. P., Shabbir, A., Silva, R., Adamson, D., Shu-Sheng, L., and Furlong, M. J. 2012. Estimating the economic cost of diamondback moth resistance to insecticides in Australia. Crop Protection 38: 30–36.

Acknowledgements:
I would like to acknowledge the Grimm Family for providing financial support for article development.
I would like to acknowledge Dr. Ian Grettenberger (University of California at Davis) for providing images for this article and both Dr. Grettenberger and Dr. Jeana Cadby (Western Growers) for input on the basic concepts presented.

