Back in 2004, and again in recent years, there were concerns by fig growers mainly in Madera and Merced counties about an excessive killing of major branches of their fig trees (Figure 1). Visits to some orchards back then and recently indicated that indeed they had a major problem. Initial close examinations of the dead branches showed symptoms which were similar to another disease: branch wilt of walnut.

The bark of dead fig branches had cracks and one could easily remove large pieces of the bark, exposing the woody tissues underneath which were covered by a black powder. Rubbing this black powder with your finger could easily remove masses of it (Figure 2). The inner surface of the broken and removed bark pieces were also black due to these powder masses. A lot of trees had many dead major branches while others had one or two dead along with other branches bearing chlorotic and thin canopy, distinct from the green and dense canopy of healthy branches.

Pathogen Activity
To collect samples, we cut some of the symptomatic branches close to the interface of dead and alive-looking (green) tissues. We noticed that in a cross section, the dead woody tissues were delineated from the healthy tissues by a dark brown line while the living woody tissues were white (Figure 1).
Slices of these woody tissues from the branches were taken, isolations were made in the laboratory and a fungus known to be a pathogen of woody tissues was consistently recovered. The name of this pathogen is Neoscytalidium dimidiatum, which is a new taxonomic name of Hendersonula toruloidea fungus, which represents the pathogen first reported to cause the branch wilt disease of walnut.
Checking the literature, the same fungus under a different name (i.e. Nattrassia mangifera) was reported in 1945 on commercial figs in California as well as on Ficus religiosa and Ficus bengalensis, causing dieback and trunk cankers. In addition to walnut branch wilt, which is a common disease of walnuts grown in the San Joaquin Valley, the same fungus was reported in causing branch wilt and dieback of poplar, eucalyptus and mango. In other reports, we found out that this pathogen can cause killing of major branches of walnut, ash trees and grapefruit. More recently, it has been reported causing cankers and hull rot of almond. On fig shoots, the pathogen grows and infects injured bark (i.e. mechanical wounds, wounds by hail or sunburn), invades the woody tissues and kills the branch. When the branch is killed, it dries and usually the bark cracks, exposing large masses of black spores. These are not true spores but are small segments of mycelia that become black as the tissues dry up and break down into small pieces, producing a layer of black powder under the bark. The fungus also produces pycnidia that protrude through small cracks of the bark (See Figure 3). However, it is the spores produced in masses by the breaking mycelia called arthrospores. that can be spread readily by air and/or splashing rain and can cause infections of pruning wounds and other injuries of branches.

Survey of Affected Areas
Before doing pathogenicity studies with the Neoscytalidium fungus, we wanted to make sure that this fungus was found frequently throughout the area where fig trees showed similar symptoms to the ones we initially observed in Madera County. Therefore, a survey of 16 fig orchards with branch dieback symptoms, representing all the major fig varieties (Black Mission, Calimyrna, Conadria and the male trees (Roeding and Stanford caprifig varieties)) was done in Fresno, Madera and Kern counties. Neoscytalidium was isolated in all of these orchards.
Limb and branch samples from the majority of these orchards had 60% to 100% Neoscytalidium, while three had 7% to 11%, and two 26% to 32%. In 12 of these orchards, a second pathogen, Phomopsis spp., was isolated along with Neoscytalidium in the first year of the survey. Phomopsis sinarencis has been reported in California causing an epidemic on Kadota figs back in 1935 and in other countries as an important fig canker pathogen. By the third year of this survey, less Phomopsis was isolated, and, very recently, almost none was isolated, probably because the very susceptible Kadota variety is rarely now planted in California. Phomopsis is known as a pathogen fungus associated with canker diseases in many other crops around the world, but more investigations are needed to figure out its role in fig limb dieback.
Differences in Susceptibility
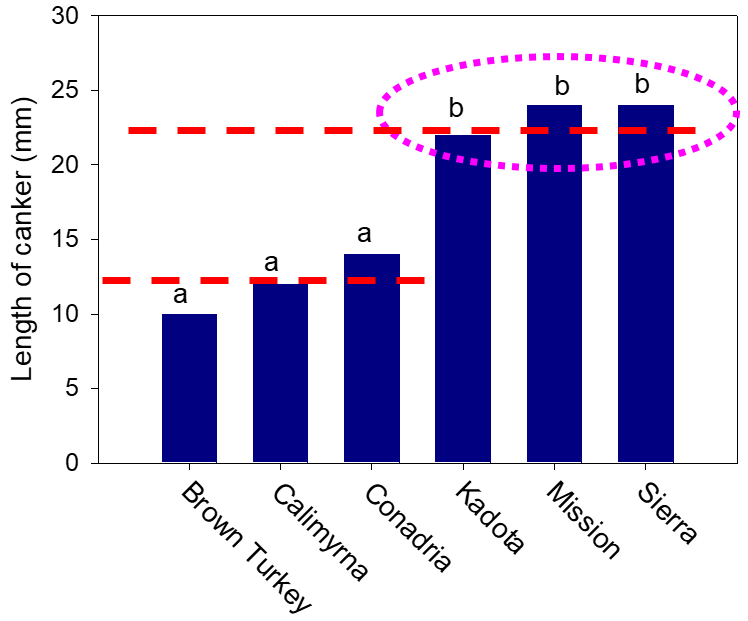
To determine if there were any differences in susceptibility to the limb dieback pathogen, we inoculated six cultivars directly in the field. We found that three months after inoculations, the cultivars Kadota, Black Mission and Sierra developed twice as long canker size than the cultivars Brown Turkey, Calimyrna and Conadria (Figure 4). Growers also reported that they see the problem to be more severe in Black Mission than other cultivars. Inoculations of six cultivars showed that Neoscytalidium is a plant pathogen that likes high temperatures. For instance, it cannot grow below 50 degrees F; its optimum temperature for growth is 90 to 95 degrees F, and it can even grow at 104 degrees F to some extent. Therefore, this fungus likes hot summer temperatures and prefers to infect sunburned branches and pruning wounds.
In experiments, we inoculated shoots of fig of different ages, including current growth (green) shoots, one-year, two-year and three-year-old shoots, by wounding and inoculating with either a mycelial plug or a spore suspension. Interestingly, the three-year-old shoots developed almost three-fold larger cankers than the cankers on current and the one-year-old shoots. This suggests that larger cuts in the field during pruning seem to be more susceptible to infection than cuts made in current or one-year-old shoots. Also, inoculations done in May, June and July resulted in larger cankers than those done from August to November. ‘
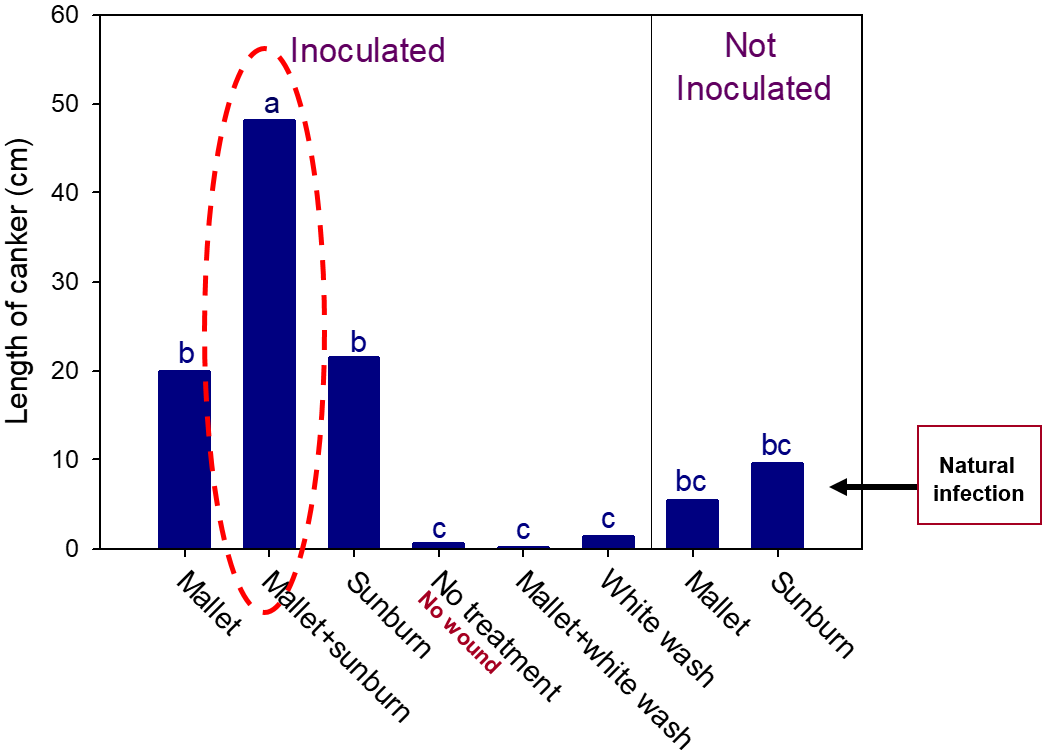
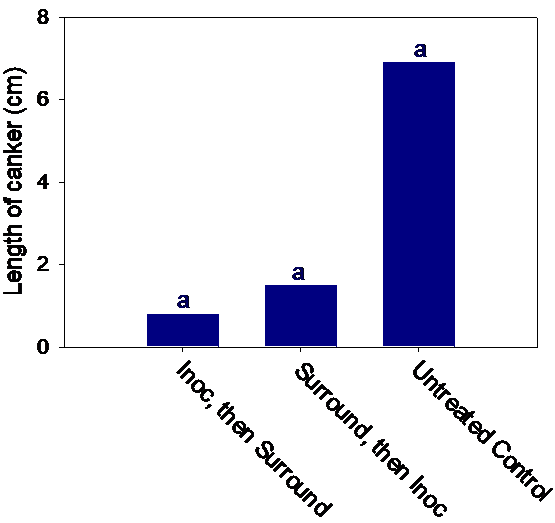
When we compared infection on pruning wounds done in winter vs. those done in summer, pruning wounds in the summer developed almost threefold larger cankers than those done during winter months. Therefore, it is recommended that pruning of figs should be done in winter when pruning wounds seem to be less susceptible. Figs can be protected from infections of the branch wilt pathogen if shoots are painted with whitewash to protect them from sunburn. Applying Surround® on shoots also protected the shoots from infection, and this is recommended to become a routine practice by fig growers. Figure 5 shows results of inoculation experiments done following various treatments in the field and artificial inoculation with the pathogen.
Although wounding by only mallet or only sunburn resulted in larger canker than the non-inoculated, un-wounded/un-treated shoots, the shoots that were damaged by mallet wounding and sunburn at the same time resulted in the longest cankers. White wash or spraying with Surround protected the shoots even after wounding with mallets and inoculation (Figure 5 and 6). Therefore, pruning that exposes the shoots to sunburn and or any other type of wounding should be avoided, and spraying with Surround will help protect the fig shoots from the limb dieback pathogen.
The authors thank the California Fig Institute for funding this research and a number of fig growers who allowed us to sample their orchards.