Microbial and botanical biostimulants have been used for promoting plant growth and to help plants withstand various pests, diseases or environmental stressors. While macro and micronutrients are necessary for plant growth and optimal yields, biostimulants play multiple roles by increasing the bioavailability of nutrients, improving nutrient and water absorption, and protecting plants from pestiferous organisms, either through direct antagonism or by triggering plants’ defense mechanisms (Berg, 2009). In addition to improving health and yields, biostimulants are also known to increase nutritional quality (Parađiković et al., 2011; Fierentino et al., 2018).
Multiple field studies in California demonstrated the potential of biostimulants and soil amendments in improving yields in tomato (Dara, 2019a; Dara and Lewis, 2019) and strawberry (Dara and Peck, 2018; Dara, 2019b). As the knowledge of biostimulants and their potential for sustainable agriculture is expanding, there has been a steady introduction of biostimulant products in the market warranting additional studies. A study was conducted to evaluate the potential of different biostimulant materials on strawberry growth, health and fruit yields.

Methodology
This study was conducted in an experimental strawberry field at the Shafter Research Station during 2019-2020. Cultivar San Andreas was planted on October 29, 2019. No preplant fertilizer application was made in this non-fumigated field, which had both Fusarium oxysporum and Macrophomina phaseolina infections in the previous year’s strawberry planting.
Each treatment was applied to a 300-foot-long bed with single drip tape in the center and two rows of strawberry plant on either side. Sprinkler irrigation was provided immediately after planting along with drip irrigation, which was provided one or more times weekly as needed for the rest of the experimental period. Each bed was divided into six 30-foot-long plots representing replications with 18-foot buffers in between. This study included both biostimulant and nutrient supplements, but this article presents data from the biostimulant treatments only. Treatments were applied either as fertigation through the drip system using a Dosatron, or sprayed over the plants with a handheld garden sprayer.
Treatments
The following treatments were evaluated in this study:
Grower Standard (GS): Between November 6, 2019 and May 9, 2020, 1.88 qt of 20-10-0 (a combination of 32-0-0 urea ammonium nitrate and 10-34-0 ammonium phosphate) and 1.32 qt of potassium thiosulfate were applied 20 times at weekly intervals through fertigation. This fertilizer program was used as the standard for all treatments except for the addition of biostimulant materials.
GS + Abound: Transplants were dipped in 7 fl oz of Abound (azoxystrobin) fungicide in 100 gal of water for four minutes immediately before planting. Transplant dip in a fungicide is practiced by several growers to protect from fungal diseases and is considered as another standard in this study.
GS + Str10 + Rhizolizer: Str10 (Wickerhamomyces sp.) was applied at 5 fl oz/ac with molasses at 10 fl oz/ac immediately after planting and Rhizolizer (Trichoderma harzianum and Bacillus amyloliquefaciens) at 3 fl oz with a food source blend at 10 fl oz two weeks after Str10 application through the drip system. The same pattern was repeated starting from mid-February 2020. From February to May, 6 fl oz/ac of Rhizolizer was applied with 20 fl oz/ac of food source once a month. Str10 is an unregistered product with yeast that is expected to help with nutrient uptake and phosphorous mobilization for improved plant vigor and yield. Rhizolizer is expected to solubilize soil nutrients and improve crop growth and yield.
GS + ON-Gard: 32 fl oz of ON-Gard was applied every two weeks through the drip system from planting until canopy development and then sprayed in 50 gpa. ON-Gard is expected to increase the nutrient use efficiency and decrease abiotic stress to the plants.
GS + ON-Gard + RootShield Plus: 32 fl oz of ON-Gard (soy protein-based) was applied every two weeks through the drip system from planting until canopy develops and then sprayed in 50 gpa. RootShield Plus WP (T. harzianum and T. virens) was also applied at 2 lb/ac through drip immediately after planting with 1 lb/ac at the end of November and again at the end of December 2019. RootShield is a biofungicide expected to protect strawberry from phytopathogens and improve water and nutrient uptake.
GS + CropSignal: CropSignal was applied at 10 gpa six days before planting and at 5 gpa 30 days after transplanting through the drip system. CropSignal is a carbon-based nutrient formula containing botanical extracts along with cobalt, copper, manganese and zinc, and is expected to support the growth and diversity of beneficial aerobic soil microbes for improved soil structure, water retention, nutrient cycling and plant protection.
Parameters observed during the study included canopy growth (area of the canopy) in January, February and March; first flower and fruit count in January; leaf chlorophyll and leaf nitrogen (with chlorophyll meter) in January, February and May, fruit sugar (with refractometer) in March and May; fruit firmness (with penetrometer) in March, April and May; severity of gray mold (caused by Botrytis cinereae) and other fruit diseases (mucor fruit rot caused by Mucor spp. and Rhizopus fruit rot caused by Rhizopus spp.) 3 and 5 days after harvest (on a scale of 0 to 4 where 0=no infection; 1=1-25%, 2=26-50%, 3=51-75% and 4=76-100% fungal growth) in March and May; sensitivity to heat stress (expressed as the number of dead and dying plants) in May; and fruit yield per plant from 11 weekly harvests between March 11 and May 14, 2020. Data were analyzed using analysis of variance in Statistix software and significant means were separated using the Least Significant Difference test.
Results and Discussion
The impact of treatments varied with different measured parameters. The interactions among plants, beneficial and pathogenic microorganisms in the crop environment, the influence of environmental factors and how all these biotic and abiotic factors respond to various biostimulant inputs can be very complex. The scope of this study was only to measure the impact on growth, health and yield parameters, not to investigate those complex interactions.
The canopy size does not always correspond with yields but could be indicative of stresses and how the plant is responding to them in the presence of treatment materials. Plants in some treatments had significantly larger canopy size in January and February, but the grower standard plants were significantly larger than the rest by March. Leaf chlorophyll and nitrogen contents were significantly different among treatments only in January where the grower standard plants had the lowest and the plants that received CropSignal had the highest. When the counts of the first onset of flowers and developing fruits were taken in January, plants that received ON-Gard alone had the highest number followed by the CropSignal and Abound treatments.
A similar trend was also seen for the average fruit sugar content. There was no statistically significant difference in the average fruit firmness among the treatments, although the value was numerically higher for the fruits in the CropSignal treatment. Severity of the gray mold, which occurred at low levels during the observation period, did not statistically differ among the treatments, but it was numerically higher in fruits from the grower standard plots. However, the severity of other diseases was significantly different among various treatments with the highest level in fruits from the grower standard and the lowest in fruits from plants that were treated with Abound. Temperatures were unusually high during the last week of May, and several plants exhibited heat stress and started to die. The number of dead or dying plants on May 28 was the highest grower standard and the lowest in Str10 + Rhizolizer and Abound treatments, although the differences were not statistically significant.
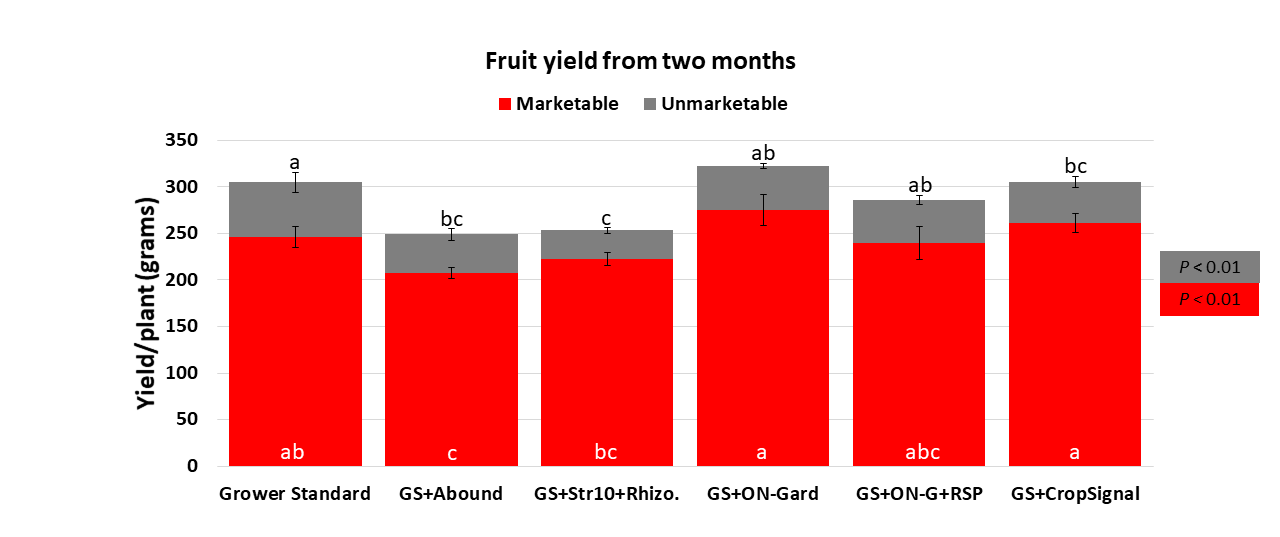
treatments, which has also been seen in some earlier strawberry studies.
Compared to the grower standard, marketable fruit yield improvement was seen only in ON-Gard and CropSignal treatments. However, marketable fruit yield was higher in all treatments compared to the grower standard with Abound. Sometimes, natural balance of the nutrients, organic matter and microbial community in the soil might result in optimal yields in the absence of pathogens or other stressors. However, it is very common to use fungicidal treatments or add biological or synthetic amendments to protect from potential threats and improving yields. These results help understand the impact of various biostimulant materials and warrant the need to continue such studies under various environmental, crop and soil conditions.

References
Berg, G. 2009. Plant-microbe interactions promoting plant growth and health: perspectives for controlled use of microorganisms in agriculture. Appl. Microbiol. Biotechnol. 84: 11-18.
Dara, S. K. 2019a. Effect of microbial and botanical biostimulants with nutrients on tomato yield. CAPCA Adviser, 22(5): 40-45.
Dara, S. K. 2019b. Improving strawberry yields with biostimulants: a 2018-2019 study. UCANR eJournal of Entomology and Biologicals. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=31096.
Dara, S. K. and D. Peck. 2017. Evaluating beneficial microbe-based products for their impact on strawberry plant growth, health, and fruit yield. UCANR eJournal of Entomology and Biologicals. https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=25122.
Dara, S. K. and D. Peck. 2018. Evaluation of additive, soil amendment, and biostimulant products in Santa Maria strawberry. CAPCA Adviser, 21 (5): 44-50.
Dara, S. K. and E. Lewis. 2019. Evaluating biostimulant and nutrient inputs to improve tomato yields and crop health. Progressive Crop Consultant 4(5): 38-42.
Fiorentino, N., V. Ventorino, S. L. Woo, O. Pepe, A. De Rosa, L. Gioia, I. Romano, N. Lombardi, M. Napolitano, G. Colla, and Y. Rouphael. 2018. Trichoderma-based biostimulants modulate rhizosphere microbial populations and improve N uptake efficiency, yield, and nutritional quality of leafy vegetables. Frontiers in Plant Sci. 9: 743.
Parađiković, N., T. Vinković, I. V. Vrček, I. Žuntar, M. Bojić, and M. Medić-Šarić. 2011. Effect of natural biostimulants on yield and nutritional quality: an example of sweet yellow pepper (Capsicum annuum L.) plants. J. Sci. Food. Agric. 91: 2146-2152.
(The author would like to thank BioWorks, Inc., Fauna Soil Production and Locus Agricultural Solutions for the financial support of the study and Marjan Heidarian Dehkordi and Tamas Zold for their technical assistance.)